Recently, a study entitled The CRL4‑DCAF13 ubiquitin E3 ligase supports oocyte meiotic resumption by targeting PTEN degradation was pressed online in the Cellular and Molecular Life Sciences. This work was from the Heng-Yu Fan Lab at Life Sciences Institute, Zhejiang University. The core of this work is identifying the mechanism of DCAF13 as a CRL4 adaptor promoting follicle development and oocyte maturation. Mechanistically, DCAF13 maintains the activity of phosphatidylinositol 3-kinase (PI3K) signaling by targeting the polyubiquitination and degradation of PTEN, a lipid phosphatase that inhibits PI3K pathway as well as oocyte growth and maturation.
In the post-natal ovaries of mammalian species, fully grown oocytes in follicles are arrested at the diplotene stage of the first meiotic prophase, which is also known as the germinal vesicle (GV) stage. With the stimulation of pituitary gonadotropins in mid-estrus, meiosis resumes with the signature of GV breakdown (GVBD) followed by chromatin condensation. During the prometaphase of meiosis I (Pro-MI), the spindle is assembled accompanied by the capture of condensed chromosomes. When all chromosomes align at the spindle middle plate in the metaphase of meiosis I (MI), the meiotic cell cycle progresses to anaphase I (AI), followed by chromosomes segregation and the exclusion of the polar body-1 (PB1). Then the ovulated oocytes entered another cell cycle arrest at the metaphase of meiosis II (MII) until fertilization. As previously reported, the PI3K/AKT signaling pathway is essential for promoting follicle development and oocyte meiotic resumption in mammals. PTEN, a major negative regulator of the PI3K pathway, needs to be degraded in fully grown follicles. On the contrary, lacking PTEN in oocytes causes the entire primordial follicle pool to become activated. Subsequently, all primordial follicles become depleted in early adulthood, causing premature ovarian failure (POF). Nonetheless, the mechanism underlying the timely degradation of PTEN in oocytes remains an unanswered question in female reproductive biology.
Heng-Yu Fan team previous has reported that DCAF13, as a novel CRL4 adaptor, is a highly expressed zygotic gene in both murine and human embryos. Dcaf13 knockout murine embryos are arrested at the morula stage (Zhang, et al, EMBO Journal, 2018). In order to further clarify the function of DCAF13 in follicles and oocytes, Heng-Yu Fan team specifically deletes DCAF13 in oocytes, which leads to the arrest of oocyte development in the NSN configuration, follicular atresia, premature ovarian failure, and female sterility. The DCAF13 deficiency results in pre-rRNA processing defects and causes a ribosome assembly disorder (Zhang, et al, Cell Death & Differentiation, 2018). In the following study, the group investigates the meiotic maturation process of Dcaf13-deleted oocytes. They find CRL4BDCAF13 E3 ligase targets PTEN for polyubiquitination and degradation during meiotic resumption, which can improve the activation of AKT that is involved in CDK1 activation. Dcaf13 knockout in oocytes causes accumulated PTEN and impairs PI3K pathway. As a result, most Dcaf13 null oocytes are arrested at the prometaphase I. In addition, insufficient accumulation of CDC20 proteins also impairs the activation of the anaphase-promoting complex (APC) and prevents the metaphase-to-anaphase transition in meiosis I.
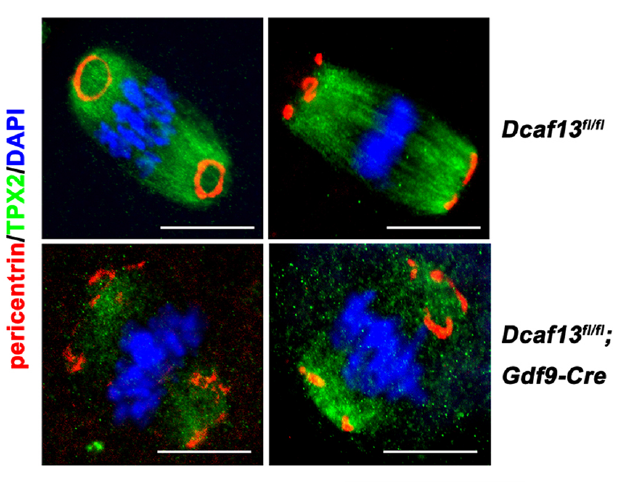
Dcaf13-deleted oocytes have defects of meiotic maturation. (Fan Laboratory, Cellular and Molecular Life Sciences, 2019)
These findings not only discover a novel factor for promoting oocyte maturation but also describe the mechanism of DCAF13 in regulating cell cycle progression. Moreover, they find CRL4DCAF13 E3 ligase targets PTEN for polyubiquitination and degradation and elucidate a long-looked-for regulation mechanism of PTEN and PI3K signaling pathway in oocytes. It has certain innovative significance for both scientific research and assisted reproduction. Dr. Jue Zhang, Dr. Yin-Li Zhang, and Graduate student Long-Wen Zhao are co-first authors of this study. Prof. Heng-Yu Fan is the corresponding author.
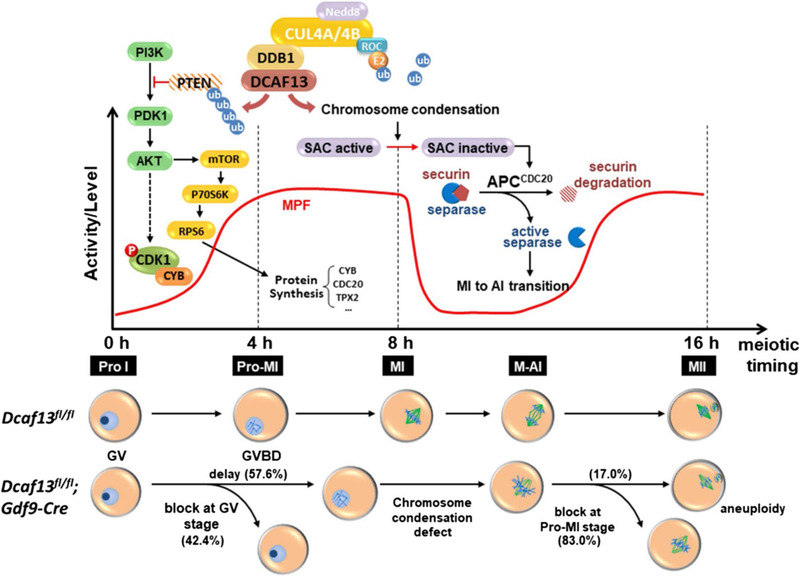
Summary of DCAF13 function in oocyte meiotic maturation. (Fan Laboratory, Cellular and Molecular Life Sciences, 2019)
Link: https://link.springer.com/article/10.1007%2Fs00018-019-03280-5